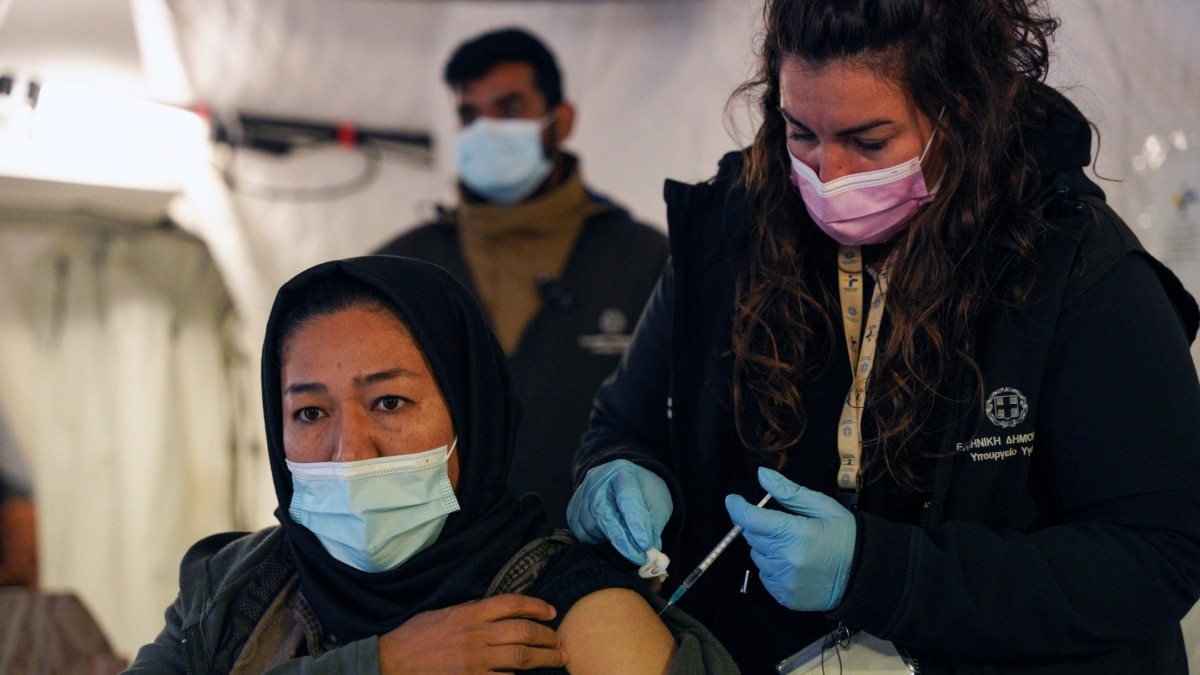
EU Health - #HealthUnion on X: "Adapted vaccines target different variants & can help maintain optimal protection as the virus evolves Today, we've authorised Moderna's BA.4/BA.5 adapted booster vaccine, following @EMA_News positive

Brianda Echevarria on LinkedIn: EMA evaluating data on booster dose of COVID-19 Vaccine Janssen - European…

European Medicines Agency Recommends Second COVID Booster For People Over 60 - As WHO Ponders Status Of COVID Emergency - Health Policy Watch
ECDC-EMA statement on booster vaccination with Omicron adapted bivalent COVID-19 vaccines | CDE Almería - Centro de Documentación Europea - Universidad de Almería

EMA evaluating data on booster dose of COVID-19 Vaccine Janssen | CDE Almería - Centro de Documentación Europea - Universidad de Almería

Repeat booster shots have immune-system risks: European Medicines Agency | World News - Hindustan Times

News - The European Commission and the Committee for Medicinal Products for Human Use at the EMA Voted Positively for mRNA Booster Vaccinations from BioNTech/Pfizer and Moderna Adapted to the Omicron Virus